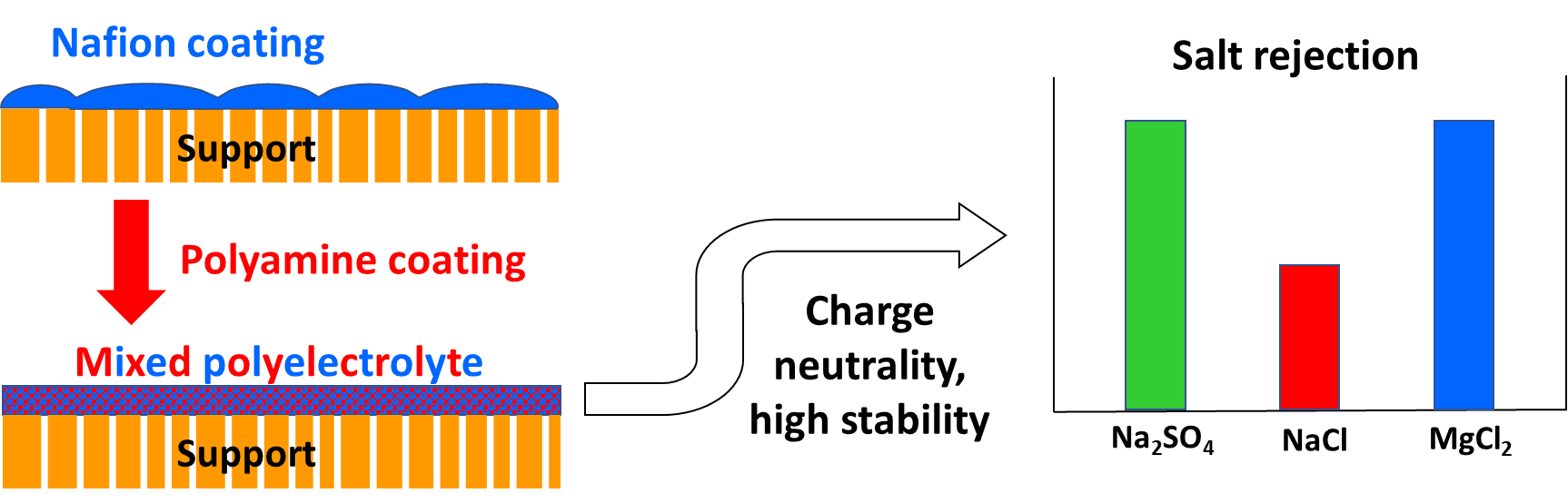Research Themes
- Next-Generation Membranes for Water-Energy-Environment Nexus
- Physics of Desalination in Membranes and Nanomaterials
- Surface Science & Engineering
“Next-Generation” Membranes for Water-Energy-Environment Nexus
Membranes are a mature field and the performance of current membranes is often good. Nevertheless, we always seek for still better performance and novel, currently unavailable, types of membranes, which requires new research and unorthodox approaches. We are working on a few such projects, such as development of biomimetic membranes based on aquaporins (water channel membrane proteins used by all living organisms), novel nanofiltration membranes that have a uniquely high permeability to salts, and membranes with improved proton conductivity for fuel cells.
Membranes for Fuel Cells (Jian Li)
A fuel cell is a device that generates electricity through an electrochemical reaction. In a fuel cell based on hydrogen as feed, hydrogen and oxygen are combined to produce electricity, heat, and water. A proton-conducting membrane is at the heart of fuel cells of this type. At present, Nafion is the benchmark membrane material that combines high chemical stability and good proton conductivity. Nafion consists of elongated polymer micelles and if they are randomly oriented then the proton conductivity is the same in all directions. However, Nafion micelles can be aligned through the plane (TP) or in-plane (IP) to enhance proton conductivity in the required direction.

We fabricate Nafion membranes using the electrospinning technique combined with the following physical folding of the prepared nanofiber mat for the orientation of the micelles.

We are focused on the study of various fundamental questions related to Nafion structure and its alignment using theoretical modeling and different fabrication methods. In particular, we have found that Nafion with elongated polymeric micelles aligned in the through-membrane direction has enhanced proton conductivity. In collaboration with Prof. Y. Tsur we explored embedding Nafion in nanopores of solid porous membranes, which served as a template for aligning Nafion micelles.

Also, we test the prepared membranes in a single fuel cell. We are attempting to develop a stable and competitive membrane with micelles aligned in the through-membrane direction.
Nanofiltration Membranes for Wastewater Recycling (Michael Zelner, Kejing Li)
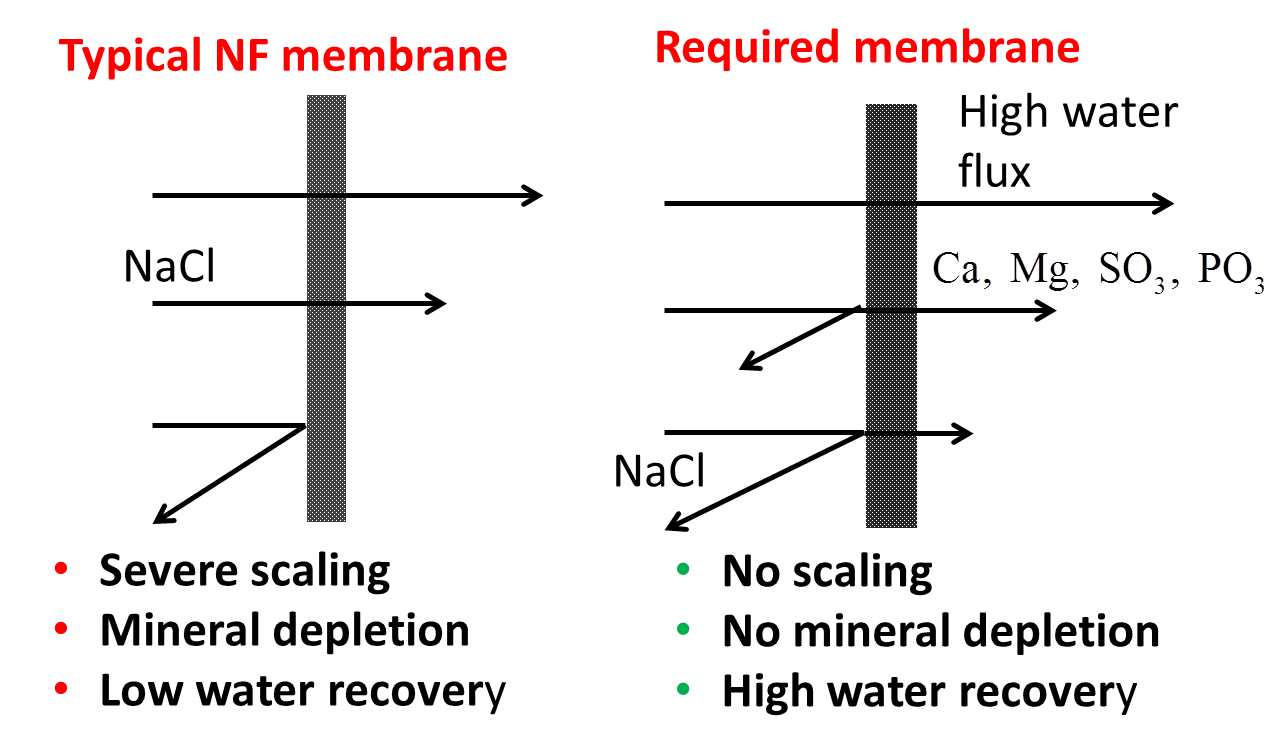
We develop novel desalination membranes with uniquely high permeation of multivalent ions, highly beneficial for water recycling. Such a novel membrane will help to reduce membrane scaling, recover more water for irrigation, and save on chemicals and fertilizers for agriculture.
Current NF membranes completely remove hardness calcium and phosphate from secondary wastewater, resulting in severe scaling and low recovery.
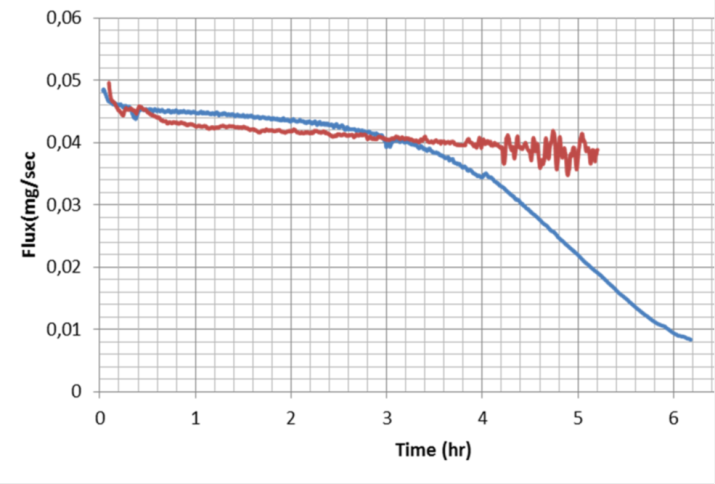
A commercial NF gets scaled and looses permeability (blue line), while a novel membrane has a better balanced Ca and PO4 rejection vs NaCl rejection and flux remains stable (red line).
Ion selectivity of nanofiltration (NF) membranes is a critical factor in NF treatment scaling-prone of multi-ion effluents, such as wastewater. When removal of multivalent ions in such effluents is unnecessary or undesired, adjusting their rejection may mitigate scaling, in which case membranes combining oppositely charged electrolytes enable a tunable selectivity towards such ions. Here we report the preparation of a new tunable and highly stable polyelectrolyte complex NF membrane prepared by successively coating an ultrafiltration membrane support with negatively charged Nafion and positively charged polyvinylamine (PVAm) layers. While Nafion thickness controls the membrane permeability, the second PVAm coating is critical for eliminating defects in the Nafion layer and readily tunes the membrane net charge and, hence, ion selectivity via PVAm concentration in the coating solution. In this manner, a membrane with good permeability, balanced charge, and, therefore, equal (“symmetric”) rejection of divalent anions and cations is obtained. The membrane performance is unaffected by exposure to 10% salt in water; such remarkable stability for a polyelectrolyte-based membrane makes it potentially attractive for the treatment of wastewater effluents and brackish water sources.
Schematic representation of preparation and properties of energy-saving Nafion-based nanofiltration membrane
Mixed-Matrix Membranes (Ruiying Li, Jiali Tang)
The selective layer of RO and NF membranes is usually slightly hydrophobic. This helps remove salts, but causes various problems, e.g. membrane foul by organics and biofilms and poorly remove some uncharged contaminants (bisphenol-A, boric acid etc.). We improve the selectivity towards contaminants and reduce propensity to fouling and facilitate cleaning by grafting a thin layer of acrylic polymer on the polyamide surface. We also insert certain molecules into polyamide structure to improve selectivity.
 principle of grafting – the coating (blue) changes surfaces properties and seals imperfections of the polyamide layer underneath
principle of grafting – the coating (blue) changes surfaces properties and seals imperfections of the polyamide layer underneath
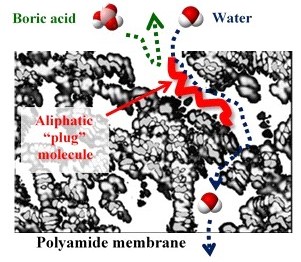 another principle – insertion of “plug” molecules into polyamide as a way to enhance selectivity
another principle – insertion of “plug” molecules into polyamide as a way to enhance selectivity
 a STEM cross-section of a modified NF membrane showing the coating (dark) covering a thin polyamide layer (bright line) on top of a porous support (light grey).
a STEM cross-section of a modified NF membrane showing the coating (dark) covering a thin polyamide layer (bright line) on top of a porous support (light grey).
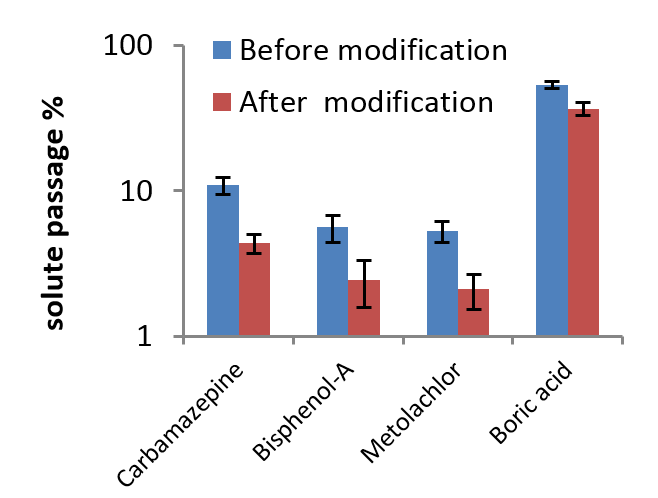 Effect of surface grafting on removal of several representative contaminants. Note the log scale for the vertical axis.
Effect of surface grafting on removal of several representative contaminants. Note the log scale for the vertical axis.
Physics of Desalination in Membranes and Nanomaterials
Understanding Desalination: “A Long and Winding Road” (Mikhail Stolov, Yaeli Oren)
In desalination membranes the salts are separated from water in a very thin (10-200 nm) selective layer made of polyamide, however, how that happens and how different ions are separated is still not well understood. We develop models and methods to study salt transport in RO and NF polyamide membranes and study the mechanism of salt and water transport using various experimental techniques, in particular electrochemical impedance spectroscopy (EIS) and cross flow filtration. We also use atomistic molecular dynamics (MD) simulations to gain better understanding of molecular mechanisms involved in salt and water transport.
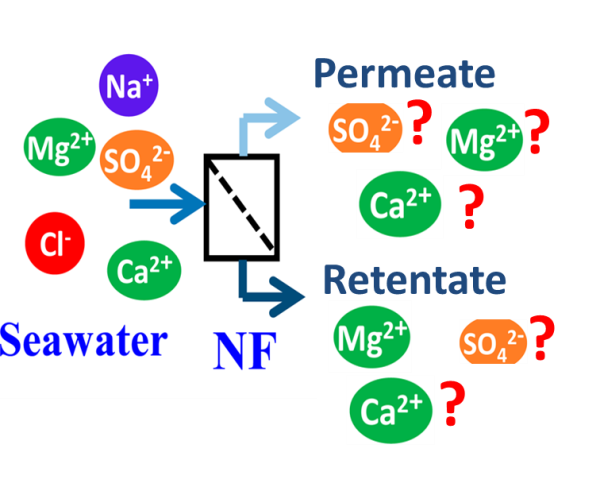
Given sea water composition, what will be the composition of permeate and retentate?
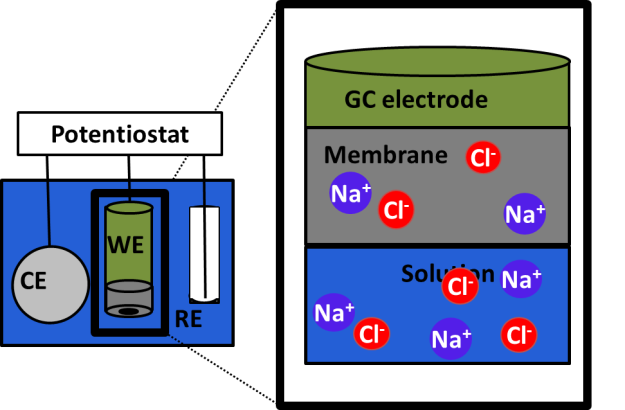 Principle of EIS: membrane conductivity supplies unique signatures of transport mechanism.
Principle of EIS: membrane conductivity supplies unique signatures of transport mechanism.
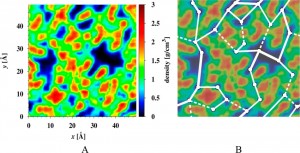
The polymer density distribution of a polyamide membrane obtained by MD and the proposed random resistor network model for water and ion transport.
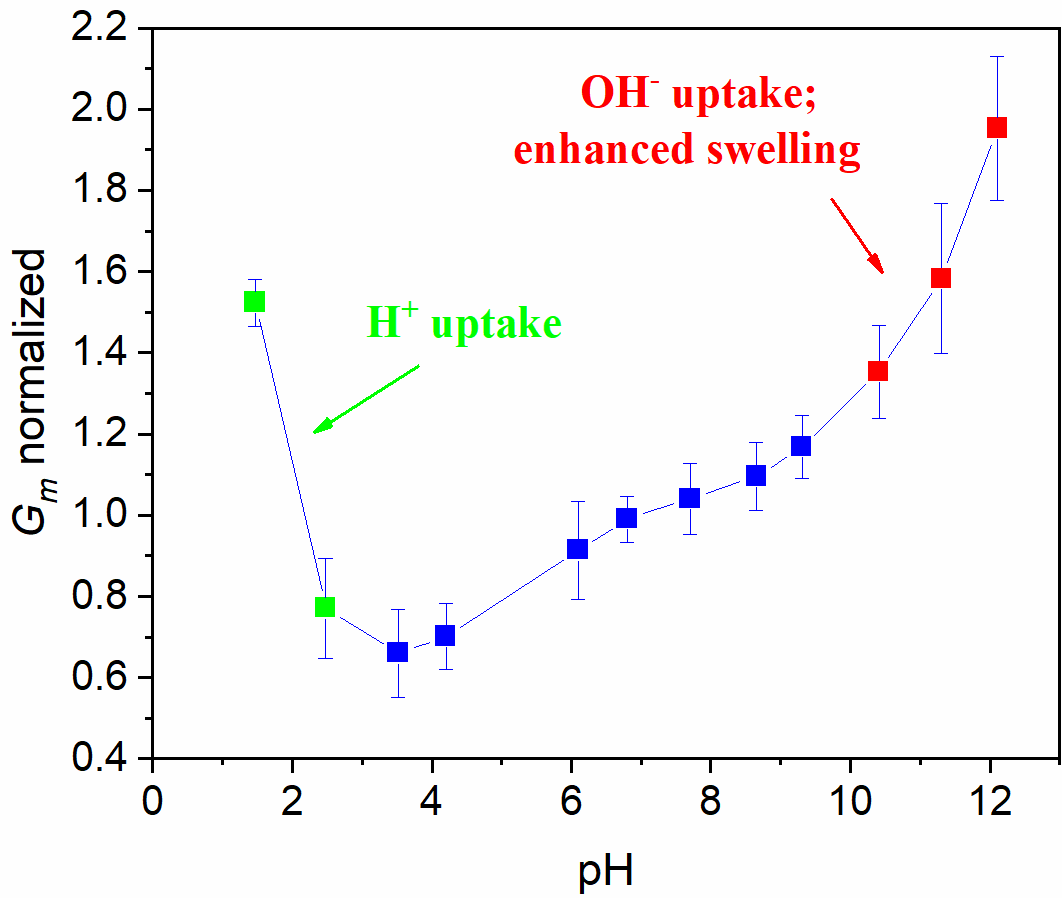
Dependence on pH of polyamide film conductivity in 1 M NaCl solution normalized to conductivity at pH 7
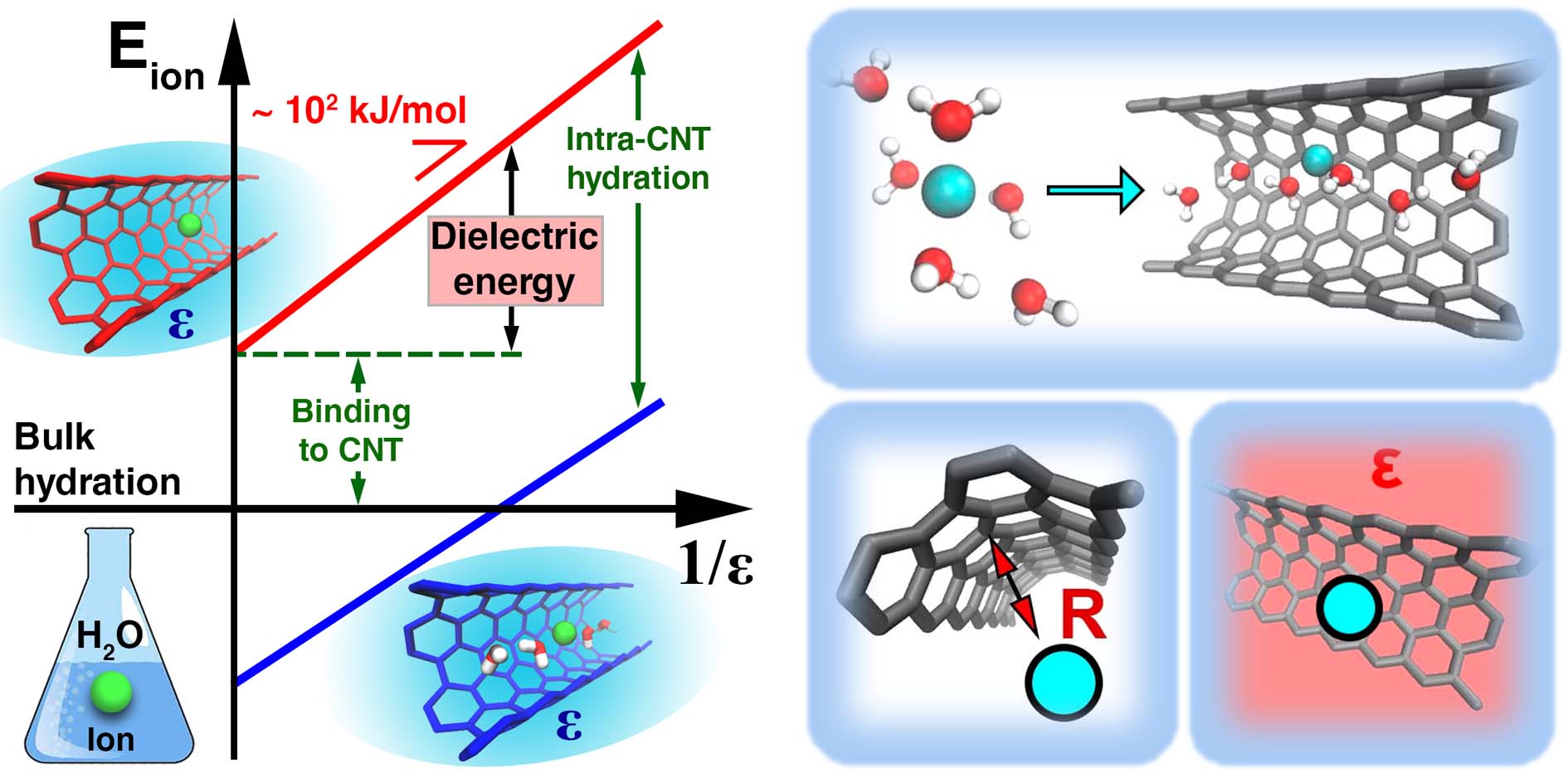
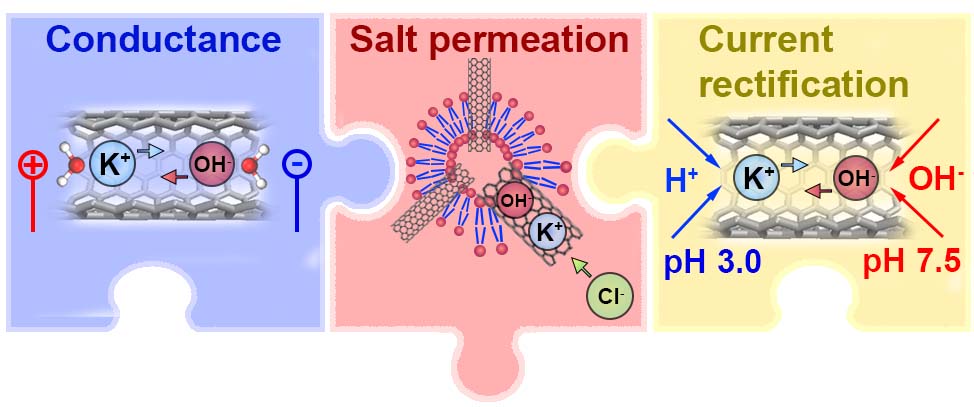
Water and Ions in Carbon Nanotube Porins: Solving the Puzzle (Vadim Neklyudov)
Sub-nanometer carbon nanotube porins (CNTPs) are unique mimics of biological water channels, yet the physics behind ion transport and selectivity in CNTPs that would rationalize experimental data remains unclear. In this research project, we fill this gap and report thermodynamic analysis of ion and water transfer to CNTP, based on ab initio calculations. The roles of the interior (intra-CNT hydration, interaction with CNT wall) and exterior (dielectric energy) are considered. The model considers the transfer of ions, as well as OH- and H+ ions present in water consequently pH dependence and salt concentration effect of conductivity are described. The proposed modeling approach may be extended to other sub-nanometer nanochannels and help design next-generation desalination and osmotic energy harvesting material and devices.
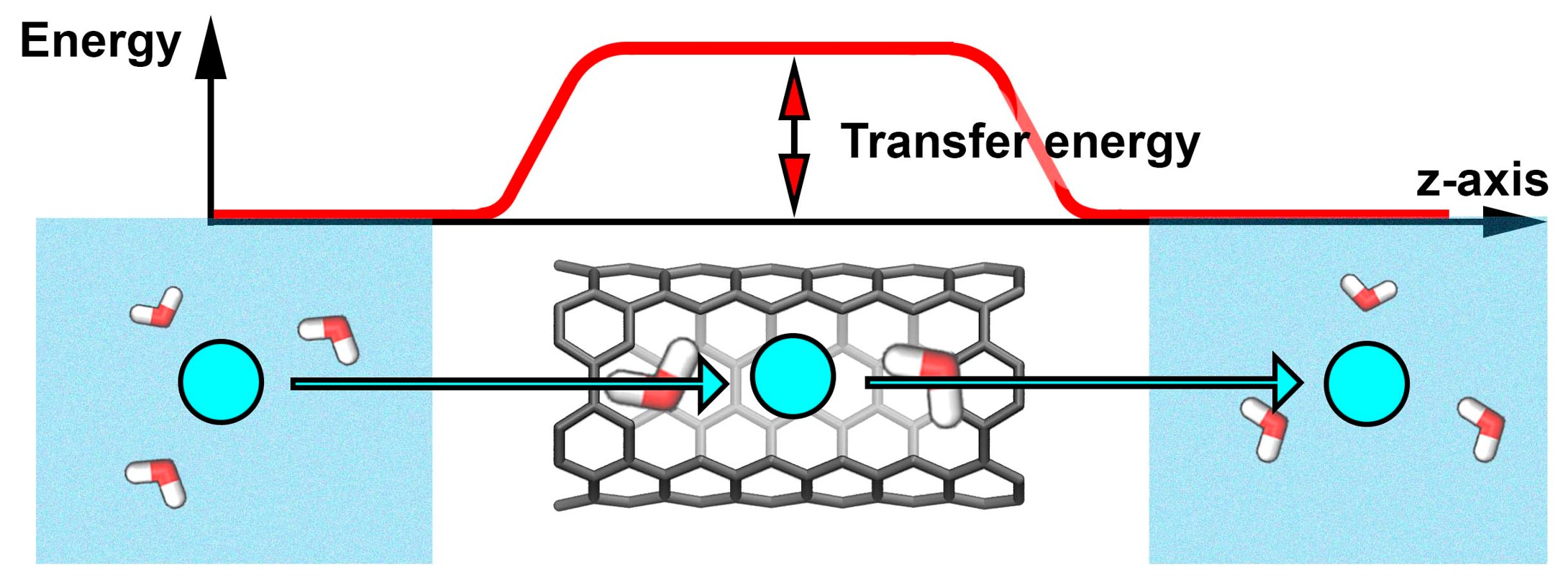
We calculated the transfer energies of ions as well as ion pairs using the density functional theory (DFT) approach.
Microfluidics as a Research Tool in Desalination (Mauricio Dantus)
Membrane technology is constantly evolving in a wide range of fields. Nevertheless, there is much room for improvement. Thus, in the field of water purification membrane manufacturing has not seen a major change for decades, undergoing only incremental improvements in permeability and selectivity, while removal of certain non-charged species, such as boron, still awaits a breakthrough. In comparison to the vast amount of work done in the field regarding charged molecules or ions, non-charged moieties remain mostly in the shadows.
In order to tackle this challenge, there is a need to measure and model the flow of mass through a membrane fully. This will help to better understand the fundamental mechanisms governing mass transfer through the membrane which is crucial for developing new and improved membranes for various fields, especially in reverse osmosis and nanofiltration systems. For this goal we want to understand the process of mass flow through the thin active layer that regulates the passage of the different components from one side of the membrane to another. We want to perform the experiments in a controlled environment without applying any external pressure, thus, learning about the mass transport phenomena through the active layer under diffusion flow regime.
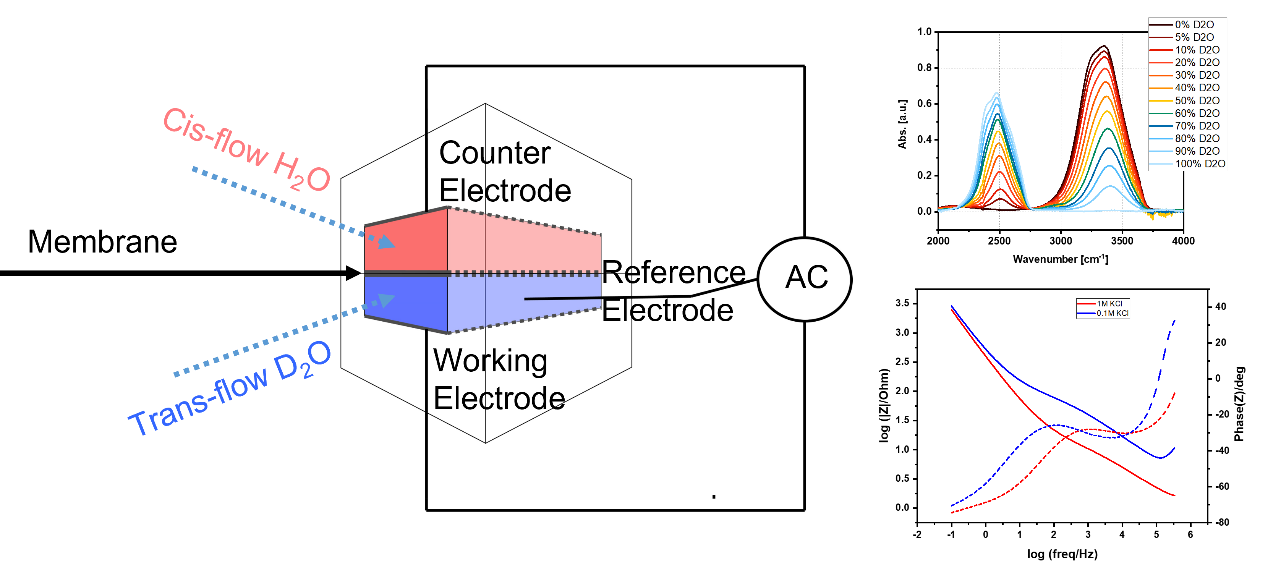
We designed a micro-fluidic device and the method to measure all the different components in our flow cell to get a full picture of the mass flow through a membrane. This device and method can help characterize many novel thin active layers that need to be characterized not only for ion related selectivity but for water transport through the membrane as well as other non-charged moieties that can be found in water-related processes. Our system includes our custom made micro-fluidic cell, conductivity measurements for salt concentration measurements, electrochemical impedance spectroscopy for in-situ charge migration measurements and infra-red spectroscopy for the water analysis. We can analyze and characterize a large number of thin films by binding them onto a surface-tunable, well-defined mechanical support. Combining the mentioned devices into one system enables us to draw a full picture of the membrane being characterized.
Cross-sectrinal scheme of our custom-made microfluidic flow cell (Left). IR spectra of H2O/D2O mixtures (Top right) and an impedance spectrum showing the difference when changing salt concentration in the solutions.
 Novel Nanofiltration Membranes for Water Recycling
Novel Nanofiltration Membranes for Water Recycling
Surface Science & Engineering
Bacteria may stick to and colonize nearly every surface in an aqueous environment by forming biofilms. In membrane processes this leads to membrane biofouling, which very difficult to predict and control. The first critical step of biofouling is deposition and adhesion of bacteria to the surface.
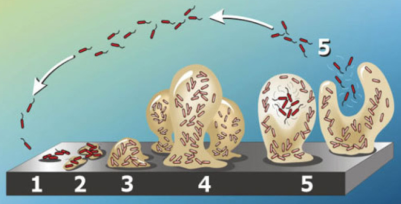 Stages of biofilm formation; stage 1 is initial deposition.
Stages of biofilm formation; stage 1 is initial deposition.
Deposition & Attachment of Particles and Living Cells on Surfaces (Lina Rosental, Aleksandr Leontev)
We study and model the mechanism of bacterial deposition experimentally (QCM-D, Parallel Plate Flow Cell (PPFC)) and theoretically, with the purpose to understand how it is related to the properties of the surface and to biofouling and how to modify surfaces to minimize their fouling. This work includes collaboration with colleagues from BGU, Technion, Germany and Korea.
 A parallel plate flow cell (PPFC) for studying deposition.
A parallel plate flow cell (PPFC) for studying deposition.
A setup for studying deposition with a PPFC mounted under microscope
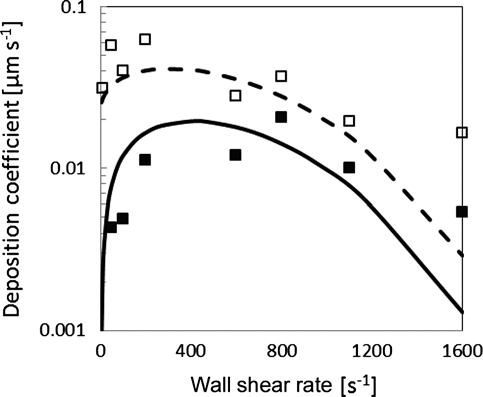
Numerical simulations can predict well bacterial deposition data
Protecting Surfaces with Soft Coating (Lina Rosental, Aleksandr Leontev)
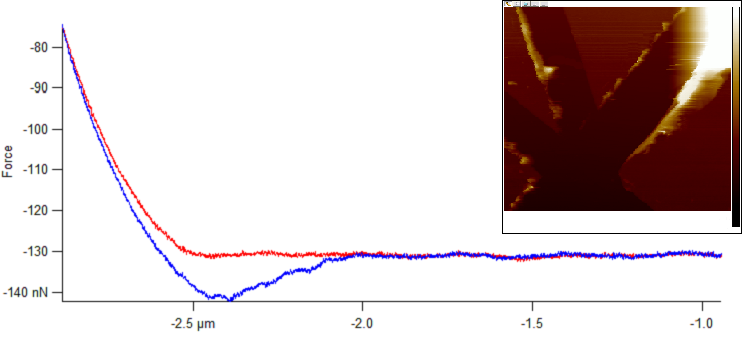
Surfaces in an aqueous environment, such as water pipes, desalination membranes or even medical implants, are prone to macromolecular and bacterial attachment. We study polymeric coatings that can protect such surfaces from contamination, with a focus on cross-linked and swellable coatings known as hydrogels. After preparing uniform and defined hydrogel coatings on glass slides, we measure their mechanical properties by atomic force microscopy (AFM). Then, we investigate and model the deposition behavior of Polystyrene particles and bacteria on the prepared coatings and on the hydrophobic and hydrophilic surfaces that are not swollen (glass slides). The deposition is analysed along a number of locations in a PPFC channel. The goal is to gain a deeper understanding of foulants interactions with the studied surfaces.
AFM scan and typical force curves of a hydrogel in water
The studied locations of deposition in a PPFC